Infra-Low Frequency Neurofeedback for Optimum Performance
By Siegfried Othmer, Ph.D. and Sue Othmer,
The EEG Institute, Los Angeles, CA, USA
February 10, 2016
ABSTRACT
A method of non-prescriptive neurofeedback is described that is based on the brain interacting with its own tonic slow cortical potential. In the absence of any explicit guidance by the clinician, the training depends entirely on the brain’s response to the unfolding signal. When this training is performed under optimal conditions in terms of placement and target frequency, there is a bias toward optimal functioning. The brain utilizes the information for its own benefit. The outcomes of the training are either comparable to or exceed expectations based on conventional EEG band-based neurofeedback. Results are shown for a cognitive skills test for an unselected clinical population.
Key words: neurofeedback, slow cortical potential, infra-low frequency training, optimum performance
INTRODUCTION
The common objective of neurofeedback is the enhancement of cerebral function and thus of organismic functional competence. This objective is not inherently deficit-focused. On the contrary, the method depends entirely on the enhancement of function that already exists. It is therefore a more organic perspective to regard neurofeedback generally as a method of achieving optimal functioning. One may even take that view if the starting point is a state of substantial dysfunction. Such dysfunctions may introduce some constraints, but the objective remains the same. Dysfunction subsides by virtue of improved function. What makes this view most appealing is that in the case of brain training with neurofeedback one is not confronted with a headroom limit. One can always do better in some respect or other. This is in contrast to much of traditional biofeedback, where the objective is to maintain good regulation of certain physiological variables, and once those objectives are achieved there is nothing more to aspire to.
The fact that most neurofeedback is currently being conducted in a clinical setting tends to shift the perspective to the remediation of deficits. Thus even the language used to communicate with clients tends to adopt that perspective. This is unsurprising for a number of reasons. The entire healthcare system is deficit-focused, and consequently clients tend to adopt that perspective as well. A somatic complaint or functional deficit will naturally focus the attention narrowly. Moreover, characterization methods used in neurofeedback tend to focus on the discernment of deficits, both to make the case for neurofeedback and to guide the training. For example, statistical parameter mapping is commonly used to determine deviations from normative behavior. Such deviations are typically linked to the diagnosis and then become targets for a ‘normalization paradigm’ in neurofeedback.
The notion that neurofeedback clinicians are actually prescribing a remedy for particular complaints is in need of a sober, cold-eyed critique. If truth be told, neurofeedback clinicians have lots of evidence that the brain may not respond as directed in training situations. Or the predicted outcome may not be achieved with the recommended protocol. Or the EEG does not change in the direction implied by the protocol. When the brain is subjected to the close-order drill that is operant conditioning, the response has greater variation than our models imply.
It is therefore more appropriate to regard the rewards and inhibits of a traditional neurofeedback protocol as a provocation or a challenge rather than as an explicit instruction. This is an easy case to make for the inhibit aspect of a training protocol. After all, the brain is merely being alerted to its transient indiscretions, and it is left to its own devices for a response. But even in the case of a targeted reward, matters are often not what they seem. Response formation depends upon the brain assigning meaning to the episodic reward, whereupon the brain is in a position to exercise all of its degrees of freedom in response. The clinician is not in good control of that process.
It has been our own experience over the years that the brain responded far too quickly to the training in many cases to be reasonably attributed to an operant conditioning response. In addition to the expected slow and gradual learning curves, we were observing state shifts and symptom relief that were surprisingly rapid and unexpected. When we first let this be known, it led to a lot of initial skepticism about our clinical findings. Our observations were not the problem, however. It was the model in terms of which such results were inconceivable. The brain was deriving more information from the signal than we thought we were providing. While we as scientist-practitioners were focusing on the operant conditioning aspects of the design, the brain was appraising the signal more comprehensively. Over time, we came to abandon the operant conditioning aspect of the training entirely, relying instead solely on the brain’s observation of its own output, as reflected in the EEG. This came about quite naturally rather than by virtue of an explicit decision, as described in the following section. The signal was now continuous, allowing the brain to experience it rather than merely to observe it. The feedback became more organic, more captivating and thus more effective, quicker to reach the goal. Once we gave up trying to dictate to the brain in an attempt to prescribe outcomes, the brain placed itself totally in charge of the process.
Once the neurofeedback process is left to the discretion of the brain in its execution, it becomes apparent that the brain utilizes all the information available to it in the cause of better regulation. The training process that has emerged is entirely analogous to the one by which the brain acquired its self-regulatory skills in the first place, during early development. We have simply given the brain the benefit of additional information, so it is the beneficiary of much more direct feedback on its own regulatory activity than is otherwise available. That in turn allows the brain’s natural proclivity toward self-optimization to be executed in a larger workspace and with greater refinement. This process is most appropriately seen in the frame of training toward optimum functioning, a matter of skill learning. Within the biofeedback field, this has traditionally been referred to as training toward mastery.
The emergence of this new kind of brain training is described briefly herein, and results are presented that make the case for the optimum functioning model. Since the results are achieved in the absence of any bias imposed on the feedback signal, the case is made that the brain utilizes the feedback signal in its own best interests, that is to say with a bias toward optimal performance.
MECHANISMS-BASED TRAINING
The original sensorimotor rhythm (SMR)/beta training approaches by Sterman and Lubar were aiming at achieving better regulatory function in first order. Sterman’s SMR training was intended to achieve better regulation of motoric excitability. In its application to seizure control, the protocol was therefore invariant, independent of the locus of any seizure focus (Sterman, 2000). The inhibits on the theta-band and high-beta band activity were intended to shore up the integrity of the rewards in the SMR-band. They were not thought to play any intrinsic training role in their own right when they were first instituted.
In Sterman’s original training of cats, there was no implication that a functional deficit was being targeted (Sterman et al., 1970). They were normal cats (or at least they were before they had electrodes implanted in their brains). The quality of their sleep improved by virtue of the training. The fact that the training effected control of chemically induced seizures was incidental to the original objective of the training, which was to investigate the effects on sleep (Sterman, 1976). In retrospect, this can be seen entirely in an optimum functioning frame.
The addition of beta1 training by Lubar was aiming at improved cognitive function in Attention Deficit Hyperactivity Disorder (ADHD) (Lubar & Lubar, 1984). His training was the first to assign an EEG shaping role to the theta-band inhibition. However, as already indicated, the inhibit function was passive in that it placed no imperative demand upon the brain, but rather merely informed it of its status of ranging outside of the bounds of good performance. The categorical remedy was the restoration of better performance, but the remedy was not narrowly specified.
The inhibit-based training was later augmented with quantitative electroencephalogram (QEEG)-based targeting, which shaped thinking more toward a deficit focus. However, matters had not been fundamentally altered. The availability of the QEEG simply enlarged the parameter space in which an appropriate inhibit strategy was to be determined. The response of the brain was still discretionary; also, the reward strategy largely remained unaffected. And even when QEEG measurements were used to inform a strategy of low-level stimulation, whether optical, auditory, or electromagnetic, these stimulations likewise served mainly in the role of a provocation or a challenge, rather than as a specification of an imperative target for the training.
The development path of the reward scheme in the Sterman/Lubar paradigm took a very different form at our hands. The availability of video feedback facilitated the presentation of the full dynamics of the training band along with the threshold crossings. In addition to this being more engaging to the trainee, it turned out to be more informative for the brain as well. Trainees responded more quickly and more profoundly, particularly with the use of bipolar montage as opposed to referential placement. It was observed that some individuals were exquisitely sensitive to the particulars of the reward frequency, and that discovery introduced the concept of the Optimum Reward Frequency, or ORF, which has guided our work ever since. With each trainee, the effort was undertaken to optimize the reward, or target frequency.
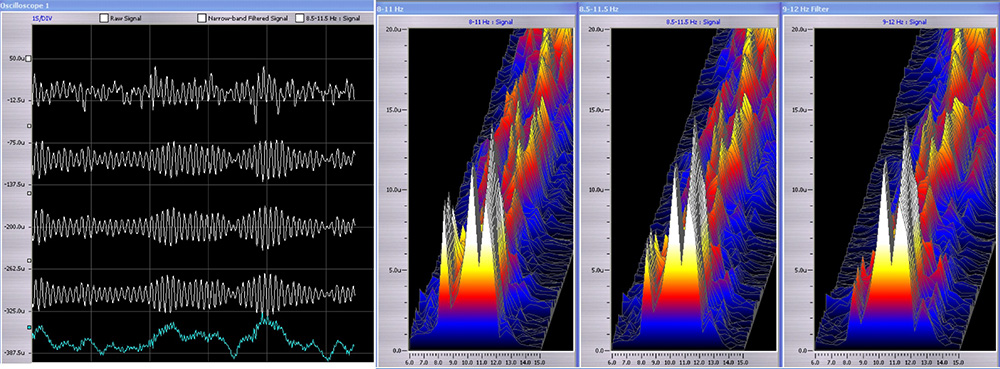
Such sensitivity to the particulars of the reward seems hard to believe when one inspects the signal at issue. An example of three training bands, separated by 0.5 Hz, is shown in Figure 1, along with the spectrals for the same bands. It is difficult to tell the difference between the signals in the time domain, and the differences likewise seem quite modest in the frequency domain. Nevertheless, sensitive responders can systematically distinguish between the different bands in training, and they can often do so very quickly, on the timescale of one to six minutes. Some individuals are sensitive to differences even smaller than 0.5 Hz. The ORFs determined for each individual are quasi-stable, changing only slowly with training if at all.
Spectral response for narrowband filter
Figure 1. Three filtered traces derived from the same source, with center frequencies spaced 0.5 Hz apart, at 9.5 Hz, 10 Hz, and 10.5 Hz, respectively. The broadband EEG is shown in the top trace, and the bottom trace shows the rectified and smoothed feedback signal for one of the three traces. The green trace shows the rectified and smoothed waveform used in feedback. The compressed spectral array is shown on the right for each of the filtered traces.
The sensitivity to target frequency was a particular issue with those afflicted with instability of brain function such as seizures, migraines, panic attacks, asthmatic episodes, and Bipolar Disorder. With individualization of the training frequency the clinical reach was extended to a much broader range of conditions, and it extended to more complex clinical manifestations. The frequency range eventually covered the entire conventional EEG range of 0.5 to 40 Hz. The distribution of target frequencies was strongly skewed toward the low end of the frequency range, and the most common target frequency was the lowest available in the software at hand: 0.1 Hz. This led to the exploration of the tonic Slow Cortical Potential as a training vehicle in 2006.
INFRA-LOW FREQUENCY (ILF) TRAINING
In the range of 0.1 Hz and below, the training is done on the basis of simple waveform-following, in which the brain merely witnesses the time course of the tonic Slow Cortical Potential (SCP), which directly reflects cortical excitability (Elbert, 1993). The target frequency is too low for conventional amplitude-based training with thresholding, so perforce operant conditioning had to be abandoned formally as the operative model. It had already become inoperative by virtue of the rapid response we had been observing, but with the abandonment of thresholding no vestige of the operant conditioning model remained. Placement was always bipolar. The dynamics of the signal then reflect the fluctuating differential cortical activation between the two active scalp sites, and the brain is observed to engage quite effectively with that information.
The gradual migration of the training to ever lower frequencies has resulted in a process that is entirely brain-based. The slowly meandering, relatively featureless signal holds no inherent interest except for the brain that produced it in the first place. And the process only gets underway once the brain recognizes its own agency with respect to the fluctuating signal. The key to that recognition must be the ongoing dynamics in the signal.
The approach differs from the well-known SCP training that operates on transient behavior in that it removes the dependence on cognitive engagement with the task (Birbaumer, 1999). In the new approach, there is no overt challenge. There is not even a requirement that the trainee be apprised of the actual signal, or even to be aware of the training procedure at all. Nevertheless, the trainee typically responds fairly promptly to the signal with a shift in arousal level, alertness, and vigilance, and such within-session response permits the optimization of the target frequency. The objective is to determine the frequency at which the trainee is optimally calm, alert, and euthymic. The training proceeds under conditions of the best-regulated state accessible to that nervous system at that moment. In the absence of felt or reported within-session response, the training is optimized on the basis of session-to-session changes.
The frequency-specificity of the conventional training carries over into the infra-low frequency region. For that reason, we refer to this approach as infra-low frequency (ILF) training. The new approach completely took over our practice in 2006, and appeared to yield better and more rapid results for all clinical conditions typically seen in a neurofeedback practice. Over time, the clinical reach was broadened to cover more challenging clients by extending the frequency range of training to below 0.01Hz. Even at such low frequencies, the dynamical aspects of the signal are sufficient to engage the brain, and to do so quite promptly.
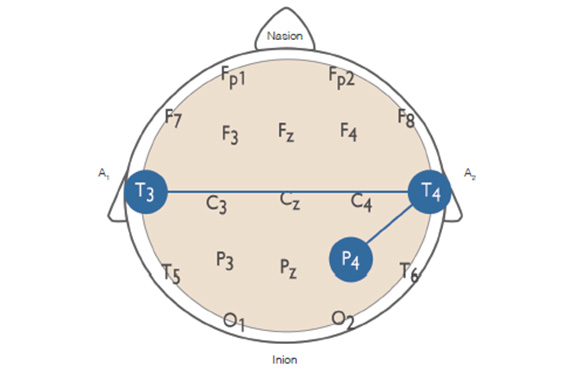
Electrode placements carried over from the higher-frequency region. These have in fact remained fairly invariant since the late nineties, but within that basic framework some tactical shifts have taken place. Brain instabilities were uniformly addressed with the inter-hemispheric placement T3-T4۱ (Othmer, 2015). This has been found to be more effective clinically than the C3-C4 placement that was more commonplace early on in the field (Quirk, 1995). It has therefore become the default starting placement for the brain instabilities.
۱T3 and T4, along with C3, C4, P4, Fp1, and Fp2 are all scalp locations designated in the International 10/20 system for electrode placement. C refers to the central cortical region, P the parietal, Fp the pre-frontal region. Figure 2 shows details of the 10-20 system.
Lateralized placements include principally T4-P4, T3-Fp1, and T4-Fp2. Whereas there was an early pre-occupation within the field with left-side placements, the extension of the work to the low frequencies has been accompanied by a shift toward right-side training as a priority. This is readily explained on the basis that core regulatory function is organized at low frequencies, and it involves right hemisphere priority. We have progressively moved toward those issues that are primary in our developmental hierarchy, issues that may not be as effectively addressed with higher-frequency training. These consist of arousal regulation, affect regulation, autonomic regulation, and interoception. These core issues are always involved in neurofeedback to some degree, but perhaps not as effectively and efficiently as with infra-low frequency training targeting the right hemisphere.
In practice, only two protocols are candidates at the outset of training, depending on whether arousal regulation or brain stability dominates in the clinical presentation. These are T4-P4 and T3-T4, as shown in Figure 2. In some instances, both are called for from the outset. Then, depending on the need, others of the four standard placements are added to the protocol. Further downstream, yet other protocols may be added for more specific purposes.
Figure 2. Starting placements for Infra-Low Frequency neurofeedback. These are either used exclusively in the first session, or they are used sequentially in the event that both are required.
This approach reflects a very clear ‘hierarchy of needs’ that becomes apparent as the various protocols are evaluated for inclusion. A kind of scaffolding applies to this process as the early protocols lay the foundation for the protocols to follow. Some of the protocols that eventually become necessary might not have been tolerated at the outset. One has the clear sense of recapitulating the person’s original developmental hierarchy and of facilitating a kind of re-ordering and of functional re-normalization.
The hemispheric division of labor plays a determinative role in the clinical decision- making process. In line with the well-known approach/withdrawal dichotomy, the right hemisphere takes primary responsibility for issues of core state regulation and of the vegetative domain. Concomitantly, cortical resting states are more broadly and intimately connected on the right side (Buckner, et al. 2008). Being primarily responsible for interoception, the right hemisphere sees to one’s sense of safety (Sridharan, et al. 2008). The left hemisphere exercises a primary responsibility with respect to executive function and engagement with the outside world. The quality of the latter is conditional on the functionality of the former. Hence the key to good left-hemisphere function actually lies with the right hemisphere.
Assessing progress in training is firstly a matter of tracking symptom severity, and secondarily one of assessing the quality of regulation broadly: the quality of sleep; of emotional regulation; of alertness and vigilance; etc. Despite the very limited targeting, the breadth of impact makes it apparent that the whole brain is affected in this process, including in particular left-hemisphere function, even before it has been explicitly targeted. Left-hemisphere functionality can be readily assessed with an instrument such as a Continuous Performance Test (CPT).
The CPT is a pressured choice reaction time test that allows one to characterize a variety of functions (Othmer, 2014). We utilize the QIKtest (www.beemedic.com), which was designed to emulate the TOVA ® (Test of Variables of Attention) (Leark, 1999). It allows a comparison of stimulus-sparse and stimulus-intensive challenges and thus explores the arousal-level dependence of functionality. By tracking the variation in performance over the session it also yields information on the stability of brain function and on the capacity to maintain vigilance under the challenge of tedium. Results were analyzed using EEG Expert (www.eegexpert.com). Initially the QIKtest relied on TOVA norms. However, as the data accumulated on a central server from a large practitioner network, it became apparent that the distributions of the discrete errors were distinctly non-Gaussian. Both omission and commission errors exhibited ‘long-tail’ (power-law) behavior. This finding invalidated the use of Gaussian statistics in the determination of normative performance. Hence the QIKtest analysis came to rely on population-based norms that were established on a database of over 50,000 records. The use of such norms is contingent only on the availability of a representative and statistically robust sample. Non-parametric statistics were used throughout. The resulting percentile scores were then converted back into equivalent standard scores by means of the conversion that applies to a Gaussian distribution, for ease of inspection and to facilitate comparisons in terms of familiar categories.
We utilize the QIKtest as an evaluation tool and progress measure with everyone who is capable of taking it, and for that reason it presents an unparalleled opportunity for the evaluation of neurofeedback with a single instrument across the entire range of functional and dysfunctional populations.
CLINICAL RESULTS
Impulsivity, as indexed by errors of commission, presents an almost ideal measure to track because the deficit lies in the functional domain and should in principle be subject to normalization. The brain has to be functional in order to be impulsive. On the other hand, the ‘normal’ range of performance leaves room for improvement. This is in contrast to the case of omission errors, where organicity plays a much larger role. Whereas non-responders may make up only about five percent of a clinical population with respect to impulsivity, they may constitute as many as 25% of the population with respect to errors of omission. Additionally, the objective of zero omission errors is commonly met by many, even in a clinical population, and is then not available for further improvement. So the inattention measure loses discrimination. For both of the above reasons, the impulsivity measure is preferred over the inattention measure to appraise neurofeedback in an optimum functioning paradigm.
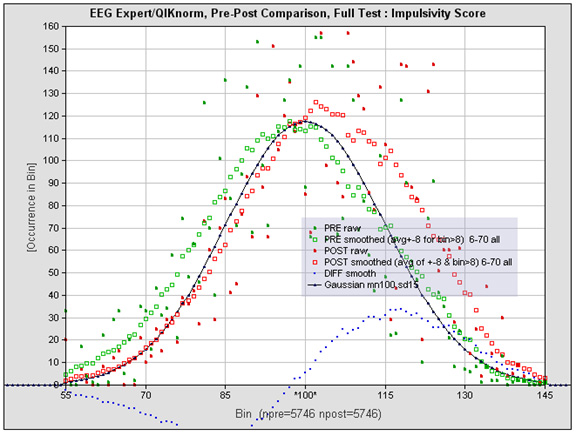
Results of the QIKtest for the impulsivity measure are shown in Figure 3 for 5,746 clients who received nominally twenty sessions of infra-low frequency neurofeedback training. The pre-training distribution is shown in green; the post-training results are shown in red. The normative distribution is shown in black. The data have been smoothed by means of near-neighbor averaging for greater clarity.
Impulsivity Score
Figure 3. The distribution of impulsivity score for a non-selected clinical population of 5,746 is shown both before and after twenty sessions of ILF neurofeedback, in green and red, respectively. The norm is shown in black. The dotted curve shows the difference pre-post. The actual distributions are not Gaussian-distributed, but have been converted to Gaussian equivalent for ease of inspection. See text for discussion.
The result of the training experience was to move the distribution above norms systematically. The deficited portion of the distribution became significantly depleted. The effect size is approximately 0.75 even for this population, for which there has been no prior selection of a deficited pool. The greatest improvement from pre-to-post is found at one standard deviation above norms. The probability of scoring two standard deviations above the mean doubled with the training, despite the fact that the score was already above norms at the outset.
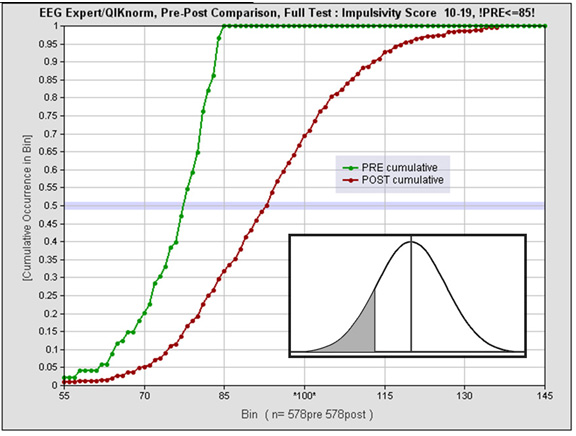
The same data set can also be used to evaluate what happens with a population in deficit with respect to impulsivity by arbitrarily limiting the sample to those who scored poorly at the outset. A cutoff of one standard deviation below norms means that we are looking at the bottom 16% of the entire distribution in terms of impulsivity. A degree of homogeneity of the sample was also sought by limiting the age range to 10-19. The resulting sample size of 578 represents the subset of the population of Figure 3 that met both criteria.
The results are shown in Figure 4 in the form of cumulative distributions. The post- training data reveal that 30% of the trainees ended up scoring above norms. The median score has improved by one standard deviation; the effect size is approximately unity for this deficited population. If the standard score of 85 is taken as the threshold for normal functionality, the training has moved two-thirds of all trainees into the functional range within twenty sessions. The pool of individuals who function in deep deficit is even more strongly depleted, with the cohort scoring below 70 (at the second percentile level) reduced by a factor of four.
Figure 4. The distributions for impulsivity score before and after training are shown for a subset of the population in Figure 2. The pool is selected in terms of age (10-19 years) and is restricted to those who initially scored more than one standard deviation below norms. See text for discussion.
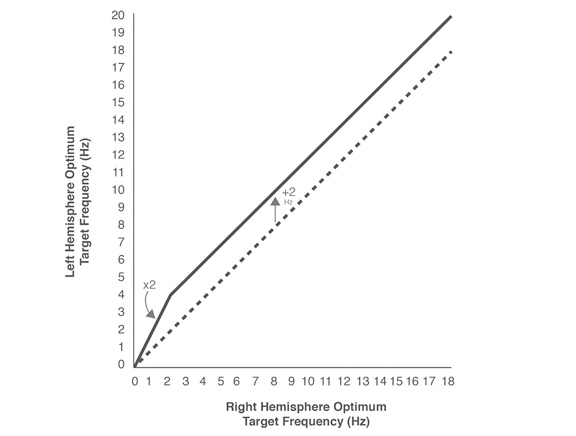
As stated earlier, the training needs to be conducted at the optimal response frequency, the ORF. A consistent finding with all placements and with all individuals is that the left hemisphere optimizes at twice the frequency of the right for all training in the ILF range (Othmer, 2013). This contrasts with the earlier finding of a difference of two Hertz between the left and the right ORFs in the conventional EEG range of frequencies. The crossover between these two regimes is at two Hertz on the right. These relationships are illustrated in Figure 5. These relationships have by now been confirmed by thousands of practitioners over a period of many years—fifteen in the case of the EEG frequency range; nine in the case of the ILF range. Exceptions to these frequency relationships have been reported by clinicians, but they tend to be quite rare.
Fig. 5: Hemispheric differences in the optimum target frequency over the entire EEG spectrum
Figure 5. A fixed relationship prevails between the optimum response frequencies in the left and right hemispheres. In the EEG range above two Hz, the left hemisphere training optimizes at two Hertz higher than the right. Below two Hz, the relationship is harmonic. The left hemisphere training optimizes at twice the frequency of the right.
DISCUSSION
The data presented encompass the entirety of such data that was available for ILF training. The sample population was unselected, consisting of the complete set of pre-post data available in our database over the timeframe of 2006 to 2014.
Consequently the data covered the time period over which the clinical method was developed from its initial beginnings in 2006. In fact, the method is still in a state of ongoing refinement. Moreover, the data were contributed by hundreds of clinicians of varying levels of experience and of clinical acumen. The clients were in many cases those for whom a bit of impulsivity would be seen as the least of their problems. The clinical focus was not on the matter of resolving impulsivity, by and large. For all the above reasons, the data of Figures 3 and 4 can be considered a valid reflection of real-world experience with ILF training in actual clinical settings with typical clients.
The significant import is first of all that the findings based on the above sample are statistically robust. Secondly, the data cement the case that neurofeedback in general, and ILF training in particular, is a method of training for optimal functioning rather than mere remediation of deficits. This follows from the fact that the training population is moved to better than normative performance, and that the relative improvement with respect to prior performance increases monotonically with score. A third observation is that many of the trainees were still on their first one or two training protocols by the twenty-session milestone, most likely targeting the right hemisphere, and yet benefit was observed for what is seen as a performance issue that strongly implicates left-hemisphere function. This makes the case for the whole-brain training effects even of lateralized placements. The possibility remains that further gains might be in prospect as left-hemisphere training is incorporated. It is also known that the benefits of training are not fully exploited by twenty sessions, particularly for those who remain in deficit at twenty sessions.
Significantly, the above results cannot be explained in terms of the placebo. First of all, there is no placebo model for the population shift above norms as seen in Figure 3. Secondly, it must be recognized that the neurofeedback training was effectively covert. Trainees were unaware of what signal they were training on, and many were unaware that they were training their brains at all, at least until they experienced the effects on their physiological state. Yet others were resolutely skeptical until they had to come to terms with their own responsiveness to the signal. Hence there was no cognitive or volitional aspect of the training process that could have mobilized the placebo response. There was no signal with which they could usefully engage even if that had been their intention. Third, the desired training effects were only available at the optimum response frequency, the ORF. That kind of specificity rules out the placebo model. Fourth, since the ORF is not known at the outset, the effects of the initial training may not accord with the desired objectives of the training, and may even be contrary to the expectations of the client. This constitutes an argument against the placebo-as-wish-fulfillment being responsible for the effects.
It is true that the protocol also incorporates an inhibit component that is not too dissimilar from other inhibit schemes that are commonly used in the field. Although the inhibit-based feedback is not obtrusive, it is not covert. Whereas a trainee is unlikely to be distracted or engaged by it, it is a readily discernible signal. Since the inhibit scheme is not substantially distinguishable from what has been standardly available in the field, it also cannot account for clinical effects that are clearly stronger than what was observed before, when similar inhibit schemes were in effect.
Moreover, attention of the trainee is not usually called to the presence of the inhibits, since these do not constitute an action item for the trainee. Questions about these subtle intrusions into the feedback signal are not usually raised until after the training effects become unambiguously apparent, at which time the client’s curiosity is aroused on the question of how that might have come about. After all, the client had not been “doing anything.” By this point, the question of a placebo effect has already been resolved with respect to that particular individual. Nevertheless, it may be argued that the inhibits are sufficient to mobilize a placebo response on the part of the trainee. To that proposition, the response is as follows:
The most compelling argument against the placebo model for ILF neurofeedback is the frequency relationship illustrated in Figure 5. The determination of the ORF is in each case based entirely on the report of the trainee on his or her own subjective experience of the training. The trainee is of course blind to the curve, and yet the client reports invariably conform to the relationships of Figure 5. This demonstrates that the clinical experience of the training is governed predominantly by what happens with the ILF component of the training rather than with the inhibits. If neurofeedback were a placebo, or even if the results were attributable mainly to the inhibit aspect of the training, then the relationship expressed in Figure 5 could not have been discovered. Instead, we have the relationship confirmed with every individual who experience both left and right-hemisphere training. Every such confirmation is an argument against the placebo model for that individual.
The above relationship points to an underlying ordering principle in the frequency domain that relates right-hemisphere priority to left-hemisphere priority. It appears that with respect to activity below two Hertz the right hemisphere has priority and the left hemisphere slaves to it at the first harmonic. At four Hertz and above, the left hemisphere has priority, and the right hemisphere follows at two Hertz behind. The imputation of left hemisphere priority in the EEG range has recently received support from the analysis of resting microstates (Pascual-Marqui et al, 2014). At the present time, there is no independent objective evidence to support the existence of the posited frequency relationships. This presents an intriguing hypothesis to be pursued.
On the above assumption, it can be argued that ILF training is concerned with those regulatory functions that are the primary burden of the right hemisphere. Since these are also the functions organized in early childhood, there is complete congruence between the hierarchy of regulation, the developmental hierarchy, and the hierarchy of organization in the frequency domain. In a comparison with our earlier higher-frequency trainings, there appears to be a clear advantage in terms of both training efficiency and outcomes if neurofeedback is begun with respect for the regulatory hierarchy, even though the data presented here do not speak to that issue.
Just as the results rule out an explanation in terms of a placebo response, the method likewise cannot be explained in terms of an operant conditioning model. The brain is merely engaged with the unfolding dynamics of the differential cortical activation, as reflected in the tonic SCP. This is closely analogous to what happens in ordinary skill learning. The brain gets feedback on the performance of the skill, but the feedback is a mere correlate of the actual activity. In the present instance the self-regulatory skill of the brain is at issue, to which the EEG—or the SCP in this case— is the operative correlate.
The mechanism appears to be the re-normalization or re-ordering of the functional coupling of our core connectivity networks, by virtue of ‘activity-mediated plasticity.’ The brain’s observation of its own state in EEG feedback propels it into novel state configurations, and every such state opens up the ‘near neighborhood’ of possible states. Every such state is susceptible to reinforcement and consolidation.
CONCLUSION
Infra-Low Frequency training is an emerging approach to neurofeedback that is intrinsically function-oriented, as opposed to targeting dysfunction. The clinical results cannot be explained on the basis of a placebo model; hence the results stand on their own, even absent validation via a placebo-controlled design. The method cannot be described in terms of the standard operant conditioning model; nor does the method rely on conscious mediation. Instead the results are explained in terms of conventional skill learning.
In its essence, the training must be understood in the optimum functioning frame, as these results are achieved without explicit guidance or micro-management by the clinician. The clinician’s role is one of discerning which ‘window into brain function’ is most salient for the brain’s burden of enhancing its own functional competence.
By working at extremely low frequencies with a right-hemisphere bias, the method addresses the foundations of our regulatory hierarchy, thus opening the door to the therapeutic relief of conditions traceable to early childhood developmental deficits or misdirections. As such, ILF training has allowed us to address challenging clinical presentations that did not yield to our earlier, higher-frequency protocols. In this work, there is a complete continuum between working with a clinical population and one concerned entirely with optimal functioning. The methods and means are the same. Consequently, ILF training has added a new dimension to the various neurotherapy approaches that have been maturing at their respective pace over the past several decades.
REFERENCES
Birbaumer, N. (1999). Slow cortical potentials: Plasticity, operant control and behavior effects. Neuroscientist, 5(2), 74-78.
Buckner, R. L., Andrews-Hanna, J. R., & Schacter, D. L. (2008). The brain’s default network: anatomy, function, and relevance to disease. Ann N Y Acad Sci, 1124, 1-38. doi:10.1196/annals.1440.011
EEG Expert, Hamburg, Germany, available at www.eegexpert.net
Elbert, T. (1993). Slow cortical potentials reflect the regulation of cortical excitability. In C. W. McCallum (Ed.), Slow potential changes in the human brain (pp. 235-251). New York, NY: Plenum.
Leark, R., Dupuy, T. R., Greenberg, L., Kindschi, C., & Corman, C. L., (1996). Test of variables of attention: Professional manual (version 7). Los Alamitos, CA: Universal Attention Disorders.
Lubar, J. O., & Lubar, J. F. (1984). Electroencephalographic biofeedback of SMR and beta for treatment of attention deficit disorders in a clinical setting. Biofeedback and Self-Regulation, 9, 1-23.
Menon, V. (2011). Large-scale brain networks and psychopathology: A unifying triple network model. Trends in Cognitive Sciences, 15(10), 483-506.
Othmer, S., Othmer, S.F., Kaiser, D.A., Putman, J. (2013). Endogenous Neuromodulation at Infra-Low Frequencies. Seminars in Pediatric Neurology, 20(4), 246-260.
Othmer, S. (2014). The Role of the Continuous Performance Test. http://www.eeginfo.com/qiktest/cpt_qiktest.pdf
Othmer, S.F. (2015). Protocol guide for neurofeedback clinicians (5th Edition). Los Angeles, CA: EEG Info Publications.
Pascual-Marqui, R. D., Lehmann, D., Faber, P., Milz, P., Kochi, K., Yoshimura, M., Nishida, K., Isotani, T., Kinoshita, T. (2014). The resting microstate networks (RMN): cortical distributions, dynamics, and frequency specific information flow. Cornell University Library. Available at http://arxiv.org/abs/1411.1949
QIKtest. Bee Medic GmbH, Kirchberg, Switzerland. Available at www.beemedic.com
Quirk, D. A. (1995). Composite biofeedback conditioning and dangerous offenders: III. Journal of Neurotherapy 1(2), 44-54.
Sridharan, D., Levitin, D. J., & Menon, V. (2008). A critical role for the right fronto-insular cortex in switching between central-executive and default-mode networks. Proceedings of the National Academy of Sciences, USA, 105(34), 12569-12574. doi: 10.1073/pnas.0800005105
Sterman, M.B. (2000). Basic concepts and clinical findings in the treatment of seizure disorders with EEG operant conditioning. Clinical Electroencephalography, 31(1), 45-55.
Sterman, M.B., Howe, R.D., & Macdonald, L.R. (1970). Facilitation of spindle-burst sleep by conditioning of electroencephalographic activity while awake. Science, 167, 1146-1148.
Sterman, M.B. (1976). Effects of brain surgery and EEG operant conditioning on seizure latency following monomethylhydrazine intoxication in the cat. Exp. Neurol., 50, 757-765.
neurofeedback neurofeedback